Every year, the FDA approves hundreds of generic drugs-and each approval saves Americans billions. But the numbers aren’t steady. Some years, savings spike. Others, they drop. Why? And what does that mean for your prescription costs?
What the FDA Tracks: Savings from New Generic Approvals
The FDA doesn’t just count how many generic drugs get approved. It calculates how much money those approvals save the system in the first year after they hit the market. This is called first-generic savings. It’s the difference between what patients and insurers paid for the brand-name version before the generic arrived, and what they pay after.
Here’s how it works: When a brand-name drug’s patent expires, the first generic version enters the market. Its price is often 70% to 90% lower than the brand. That drop isn’t just a one-time deal. It forces the original brand to lower its price too, to stay competitive. The FDA measures that entire price collapse over 12 months.
Take 2019. That was a record year: $7.1 billion saved in just 12 months from new generic approvals. Why? Because several high-cost drugs-like those for diabetes, high cholesterol, and autoimmune diseases-lost patent protection at once. When one of those drugs goes generic, it can save billions all by itself.
But then came 2020. Savings dropped to $1.1 billion. Not because fewer generics were approved. But because the drugs that lost patents that year were smaller, less expensive ones. The savings are lumpy. It’s like winning the lottery-some years you hit the jackpot, other years you get a $5 prize.
2022: A Surge in Savings
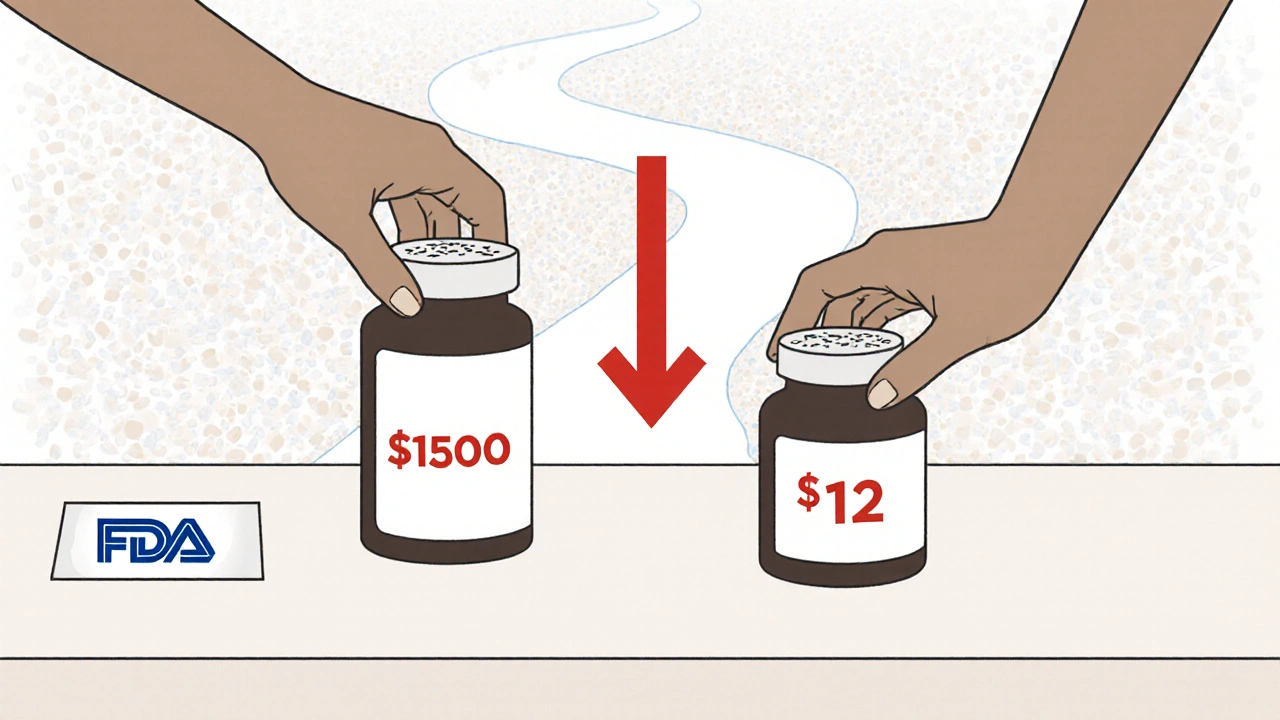
2022 was another big year. The FDA reported $5.2 billion in savings from first-generic approvals alone. That’s more than double 2021’s $1.37 billion. What changed? The FDA pointed to several approvals in "relatively large markets." That means blockbuster drugs-ones used by millions-finally got generic versions.
One example: a generic version of a widely prescribed blood pressure medication. Before approval, the brand cost $150 a month. After, the generic sold for $12. Multiply that by millions of prescriptions, and you get billions in savings.
And it wasn’t just first generics. In 2022, the FDA approved 742 generic drug applications. That’s more than two per day. Each one added to the total savings. For all generics approved that year, the total 12-month savings jumped to $18.9 billion. That’s not just one drug. That’s hundreds.
Total Savings: What the Industry Measures
The FDA’s numbers are powerful, but they only show part of the picture. The Association for Accessible Medicines (AAM) tracks something bigger: total savings from all generic drugs in use during a calendar year.
In 2023, that number hit $445 billion. That’s not just from drugs approved that year. It’s from every generic pill, injection, or inhaler used in America in 2023. That’s the real impact of generics: they’re not just new entries. They’re the default.
Think about it: 90% of all prescriptions filled in the U.S. are generics. But they make up only 13% of total drug spending. That’s the power of competition. Brand-name drugs might cost $500 a month. The generic? $15. That’s not a discount. That’s a revolution.
The savings are spread across payers:
- Commercial insurers saved $206 billion in 2023
- Medicare saved $137 billion (that’s $2,672 per beneficiary)
- Medicaid saved the rest
And it’s not just about money. It’s about access. A heart disease patient on a $300 monthly brand drug switches to a $20 generic. That’s not a savings-it’s a lifeline. Without that switch, many people would skip doses or stop taking their medicine altogether.
Who Benefits? And Who Doesn’t?
You’d think lower drug prices mean lower out-of-pocket costs for patients. And often, they do. The average generic copay in 2019 was just $6.97. Most generics cost under $20. That’s why 92% of generic prescriptions are filled at that price or lower.
But here’s the catch: not all savings reach the patient. Pharmacy benefit managers (PBMs)-the middlemen between insurers, pharmacies, and drugmakers-collect rebates from manufacturers. A 2023 Senate investigation found that only 50% to 70% of those savings actually make it to the consumer. The rest stays with PBMs, insurers, or drugmakers.
State Medicaid programs see the full benefit. California’s Medi-Cal program saved $23.4 billion in a single year. Alaska saved $354 million. The size of the savings depends on population, prescription volume, and how aggressively the state negotiates prices.
Still, for millions of Americans, generics are the only reason they can afford their meds. A cancer patient on a $10,000-a-month brand drug might pay $1,500 after a generic arrives. That’s not a 10% cut. That’s a 85% drop. That’s life-changing.
Why the Numbers Jump Around
Why did savings jump from $1.1 billion in 2020 to $5.2 billion in 2022? It’s not because the FDA got faster. It’s because of patent cliffs.
Big drugmakers hold patents for 20 years. But they often extend them with minor tweaks-new doses, new packaging, new delivery systems. That’s called "evergreening." When those tricks run out, generics flood in. And when multiple big drugs lose protection at once? Savings explode.
That’s why 2019 was so high: three major drugs lost patents. 2020 was low: only small drugs went generic. 2022? A mix of big and mid-sized drugs. The pattern isn’t random. It’s tied to when patents expire.
And it’s not just small molecules. Biosimilars-generic versions of complex biologic drugs-are starting to appear. As of August 2024, the FDA had approved 59 biosimilars. They’re harder to make, slower to approve, and more expensive. But they’re coming. And when they do, they’ll add billions more in savings.

What’s Next? The Future of Generic Savings
The FDA is working to speed up approvals for complex generics-drugs that are hard to copy, like inhalers or injectables. Their 2023 Drug Competition Action Plan targets delays caused by brand manufacturers using legal tricks to block generics.
Right now, 95% of standard generic applications are reviewed in under 10 months. That’s faster than ever. But for complex drugs, it can still take years. The FDA wants to change that.
By 2033, U.S. generic drug sales are projected to hit $131.8 billion. That’s up from $95.87 billion in 2024. Why? Because more big drugs are losing patents. More people are aging. And insurers are pushing harder for generics.
The AAM estimates that by 2028, total savings from generics and biosimilars will reach $3.9 trillion since 2014. That’s $450 billion to $500 billion a year. That’s not a trend. That’s a transformation.
Why This Matters to You
If you take any prescription drug, you’re part of this story. Even if you’re on a brand, you’re paying less because generics are forcing prices down. If you’re on a generic, you’re saving hundreds-or thousands-each year.
But you need to ask your pharmacist: "Is there a generic?" Not all doctors know. Not all pharmacies automatically switch. You have to push. And if your copay is still high, ask why. The savings are there. The question is: are you getting them?
Generic drugs aren’t just cheaper. They’re the reason millions of Americans can afford to stay healthy. And every year, as more patents expire, that safety net gets stronger.
How much do generic drugs save the U.S. each year?
In 2023, generic drugs saved the U.S. healthcare system $445 billion, according to the Association for Accessible Medicines. That includes savings from all generics in use that year-not just new approvals. The FDA also reports savings specifically from new generic approvals, which totaled $18.9 billion in 2022 from all new approvals and $5.2 billion from first-time generics alone.
Why do generic drug savings vary so much from year to year?
Savings spike when high-cost brand-name drugs lose patent protection. In 2019, several blockbuster drugs went generic at once, leading to $7.1 billion in first-generic savings. In 2020, fewer major drugs lost patents, so savings dropped to $1.1 billion. It’s not about how many generics are approved-it’s about how expensive the drugs are that go generic.
Do generic savings lower my out-of-pocket costs?
Often, yes. The average generic copay is $6.97, and 92% of generics cost $20 or less. But not all savings reach you. Pharmacy benefit managers (PBMs) often keep a portion of rebates. A 2023 Senate report found only 50-70% of generic savings flow directly to patients. Always ask your pharmacist if your generic copay can be lowered.
What’s the difference between FDA and AAM savings numbers?
The FDA measures savings from drugs approved in a given year during their first 12 months on the market. The AAM measures total savings from all generics used in a calendar year. So FDA numbers are about new entries; AAM numbers are about total market impact. Both are true-they just measure different things.
Are biosimilars saving as much as traditional generics?
Not yet. Biosimilars are generic versions of complex biologic drugs, like those for cancer or autoimmune diseases. As of August 2024, the FDA had approved 59 biosimilars. They’re harder to make, slower to approve, and more expensive than traditional generics. Their savings are growing but still small compared to the billions saved by small-molecule generics.
Can I ask my doctor for a generic drug?
Absolutely. In fact, you should. Most brand-name drugs have generic versions. Ask your doctor if a generic is available and appropriate for your condition. Even if your prescription says "dispense as written," you can still ask your pharmacist to substitute a generic if it’s allowed by law and your insurance.
How do I know if my generic drug is safe?
All FDA-approved generics must meet the same quality, strength, purity, and stability standards as the brand-name drug. They use the same active ingredients and work the same way in your body. The FDA inspects manufacturing sites, including many overseas. If your generic works for your condition, it’s safe.



Frances Melendez
November 27, 2025 AT 11:54So let me get this straight-pharma companies get 20 years to milk a drug for $500 a pill, then suddenly it’s ‘oh no, competition!’ and we’re supposed to cheer because now it’s $15? And you call that a victory? Meanwhile, my copay’s still $40 because PBMs pocket the difference. This isn’t progress-it’s a rigged game.
Jonah Thunderbolt
November 29, 2025 AT 04:37OMG YES!!! 🙌 The FDA’s numbers are *chef’s kiss* but let’s be real-this whole system is a glorified shell game. Brand names? They patent the *color* of the pill. 🤦♂️ And then they cry when generics show up like, ‘but we spent $2B on R&D!’ Bro. You spent $2B on lawyers. 💸 #GenericRevolution #PharmaIsAScam
Rebecca Price
November 30, 2025 AT 17:53It’s fascinating how we celebrate the *mechanics* of savings without addressing the moral architecture behind them. Generics aren’t just cheaper-they’re a societal contract: that health shouldn’t be a luxury. And yet, we let middlemen siphon half the benefit. That’s not inefficiency. That’s betrayal. We need transparency, not just statistics.
shawn monroe
December 1, 2025 AT 12:10Let’s talk biosimilars-this is where the real game-changer is. Traditional generics? Easy. Biologics? You’re talking about replicating a living molecule-like cloning a snowflake with a fork. The FDA’s 59 approvals are just the tip of the iceberg. By 2030, we’re looking at $20B+ in annual savings from these alone. It’s not just cost-it’s precision medicine democratized.
marie HUREL
December 3, 2025 AT 03:24I’ve been on a generic blood pressure med for three years now. It’s $12 a month. Before? $180. I didn’t even know I could ask for it. My doctor assumed I’d want the brand. I wish more people knew this was an option. It’s not just about money-it’s about dignity.
Lauren Zableckis
December 4, 2025 AT 04:34The $445 billion number is staggering, but what really hits home is the 90% of prescriptions being generics. That’s not policy. That’s culture. We’ve quietly built a system where affordability is the default. And yet, we still treat generics like second-class medicine. Time to stop the stigma.
Asha Jijen
December 5, 2025 AT 20:38Edward Batchelder
December 6, 2025 AT 00:25Every time I hear someone say, ‘I don’t trust generics,’ I want to hand them a copy of the FDA’s bioequivalence guidelines. These aren’t knockoffs. They’re identical in active ingredient, dosage, safety, and effect. The only difference? Price. And that’s not a flaw-it’s the point. We should be proud of this system, not suspicious of it.
reshmi mahi
December 6, 2025 AT 16:52laura lauraa
December 7, 2025 AT 13:06Let’s not romanticize this. The FDA’s ‘savings’ are statistical smoke and mirrors. The real cost? The psychological toll on patients who must navigate insurance labyrinths, PBM rebates, and formularies that still favor brands. And when you ask for a generic, you’re often met with condescension-‘Oh, but this one’s better.’ It’s not medicine. It’s classism, wrapped in a white coat.
Gayle Jenkins
December 7, 2025 AT 19:22Just spoke to my pharmacist yesterday-she told me my $15 generic for a cholesterol drug is actually $30 if you don’t use your insurance. Why? Because the PBM’s rebate structure makes it cheaper for them to charge you cash. So you’re literally better off paying out-of-pocket. That’s not a system. That’s a trap. Ask your pharmacist for the cash price. Always.
Frances Melendez
December 8, 2025 AT 18:22You think it’s just PBMs? Try asking your insurance company why your ‘$6 generic’ still has a $100 deductible. Or why your doctor’s office won’t even prescribe it unless you sign a waiver. This isn’t about transparency-it’s about control. And the system is designed to keep you confused, compliant, and paying.