When you take carbamazepine for epilepsy or nerve pain, you're not just taking a pill-you're managing a complex chemical dance inside your body. This drug, available in dozens of generic forms, doesn't just work on its own. It changes how your body handles other medications, sometimes dangerously so. And here's the catch: not all generic versions behave the same way, even if they're labeled the same. For many patients, switching between generics isn't just a pharmacy routine-it's a risk.
Why Carbamazepine Is a Powerhouse Enzyme Inducer
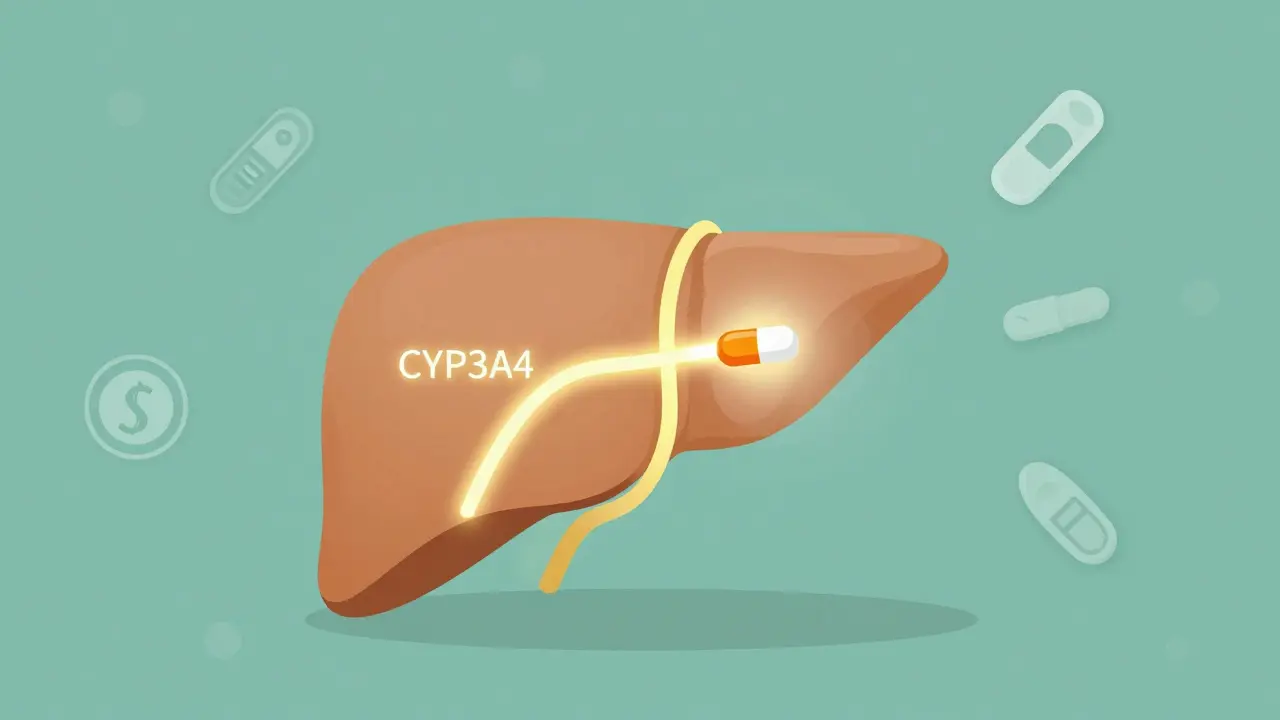
Carbamazepine doesn't just sit in your system. It actively rewires your liver's drug-processing machinery. Specifically, it turns on enzymes in the cytochrome P450 family, especially CYP3A4. This enzyme is responsible for breaking down about half of all prescription drugs you might be taking. When carbamazepine wakes it up, your body starts clearing other medications faster than normal.This isn't a slow process. Within 48 to 72 hours of starting carbamazepine, enzyme activity begins to rise. By two to three weeks, it's at peak levels. And here's what that means in real life: if you're on warfarin for blood thinning, your dose might need to go up by 30% just because you started carbamazepine. Same with cyclosporine after a transplant, or birth control pills-your body might break them down so fast they stop working.
It gets worse. Carbamazepine also induces UGT enzymes and P-glycoprotein, two more systems that push drugs out of your body. So even if one enzyme doesn't touch your medication, another might. That’s why the FDA lists over 20 drug classes that interact with carbamazepine, from antifungals to HIV meds to antidepressants. The result? A patient on multiple drugs can end up with levels of one or more drugs plummeting below effective range-without any obvious signs until something goes wrong.
The Narrow Therapeutic Window: Why Small Changes Matter
Carbamazepine’s therapeutic range is 4 to 12 mcg/mL. That’s it. Below 4, seizures may return. Above 12, you risk dizziness, nausea, double vision, or worse-like toxic confusion or heart rhythm issues. The problem? People absorb and process this drug differently. One person might need 600 mg a day to hit 8 mcg/mL. Another might hit 11 mcg/mL on the same dose. That’s a 30-40% variation in how the drug behaves between individuals.This narrow window makes carbamazepine a classic narrow therapeutic index (NTI) drug. The European Medicines Agency classifies it as such. That means even tiny differences in how a generic pill releases its active ingredient can push someone out of the safe zone. The FDA requires generics to match brand-name drugs within 80-125% of the original in terms of absorption (AUC) and peak concentration (Cmax). But those numbers are based on healthy volunteers. Real patients? They have kidney issues, liver disease, other seizure meds, or hormonal shifts-all of which alter how carbamazepine moves through their bodies.
Take a 35-year-old woman on birth control and carbamazepine. Her CYP3A4 activity is naturally 20-25% higher than men’s due to estrogen. If she switches from one generic to another-even one that meets FDA standards-her blood level might drop 15% overnight. That’s enough to trigger breakthrough seizures. Studies show women of childbearing age are 22% more likely to have seizures after a generic switch than men.
The Generic Switching Problem: What the Data Shows
You’d think if two pills are both labeled “carbamazepine 200 mg,” they’re interchangeable. But data says otherwise. A 2018 study tracked 327 epilepsy patients who switched between different generic brands. Twelve percent had treatment failure or new side effects. Nearly 8% ended up in the ER. One patient went from 7.2 mcg/mL to 4.8 mcg/mL after switching generics-same dose, same doctor, same pharmacy. Seizures went from once a month to five times a week.Why? It comes down to formulation. Immediate-release tablets are simpler, but extended-release versions-like Tegretol XR, Carbatrol, or Equetro-are trickier. They use beads or coatings to slowly release the drug over hours. If one generic uses larger beads, or a different polymer coating, the release pattern changes. In patients with gastroparesis (delayed stomach emptying), this can mean the drug isn’t absorbed properly at all. One Reddit user, a nurse with epilepsy, noticed her seizures spiked after switching to a generic made by Nostrum Pharmaceuticals. She later found out the bead size was different from her previous version.
The FDA’s own data shows 32 approved generics for immediate-release carbamazepine and 18 for extended-release. That’s 50 different formulations. Each one has a different dissolution profile, different fillers, different binders. And none of them are tested against each other-only against the original brand. So if you switch from Generic A to Generic B, there’s no guarantee B behaves like A. The FDA admits this is a blind spot. Their 2023 guidance says single-dose bioequivalence studies don’t capture what happens at steady state-especially with a drug that induces its own metabolism.
Who’s at Highest Risk?
Not everyone has problems. About 60% of patients switch generics without issue. But certain groups are far more vulnerable:- Patients with uncontrolled seizures-even a 10% drop in carbamazepine levels can trigger a seizure.
- Women of childbearing age-hormones boost CYP3A4 activity, making them more sensitive to formulation changes.
- Patients on multiple medications-each additional drug increases the chance of a dangerous interaction.
- Patients with liver or kidney disease-altered metabolism amplifies variability.
- Asian patients-those with the HLA-B*1502 gene variant have a 10-fold higher risk of Stevens-Johnson Syndrome (SJS), a life-threatening skin reaction. Screening is required before starting carbamazepine in this group.
A 2021 study in JAMA Neurology found that switching generics in women led to a 22% increase in breakthrough seizures. That’s not a small number. It’s a clinical emergency waiting to happen.

What Doctors and Pharmacists Need to Do
The American Epilepsy Society and American Academy of Neurology agree: don’t switch carbamazepine unless you have to. If a patient is stable on one brand or generic, keep them there. If a switch is unavoidable, follow these steps:- Check the manufacturer-know which company makes the current version. Write it on the prescription.
- Use DAW 1-this pharmacy code means “dispense as written.” It blocks automatic substitution.
- Monitor blood levels-check carbamazepine concentration before the switch, then again at 7-10 days and 4 weeks after. If levels change by more than 15%, adjust the dose.
- Warn patients-tell them to report new dizziness, rash, or increased seizures immediately.
Only 68% of U.S. neurologists currently use DAW 1 codes for carbamazepine. That means over a third of patients are being switched without their doctor’s knowledge. And many pharmacists don’t know the difference between the 12 different manufacturers of 200 mg carbamazepine tablets listed in the FDA’s Orange Book.
The Future: Precision Dosing and Genetic Testing
We’re moving toward a future where carbamazepine dosing isn’t guesswork. Researchers have identified 17 genetic variants that affect how the drug is processed. One common variant, CYP3A4*22, means a patient needs 25% less drug to reach therapeutic levels. Another affects how fast the drug is cleared. In pilot studies, using genetic data to guide dosing cut adverse events by 35%.The FDA’s Sentinel Initiative is tracking 500,000 carbamazepine users through 2025 to see what really happens when people switch generics. Early data will be out in early 2025. Meanwhile, the American Epilepsy Society is developing a Therapeutic Drug Monitoring Toolkit for 2024, which will include algorithms that factor in age, sex, weight, and other medications.
For now, the safest approach is simple: know your version, monitor your levels, and never assume generics are interchangeable. Carbamazepine isn’t like ibuprofen. It’s a powerful, unpredictable drug-and in the world of generics, that’s not just inconvenient. It’s dangerous.


Aisling Maguire
February 28, 2026 AT 14:28bill cook
March 1, 2026 AT 22:27Lisa Fremder
March 2, 2026 AT 14:20Justin Ransburg
March 3, 2026 AT 03:15Sumit Mohan Saxena
March 3, 2026 AT 19:11Ben Estella
March 4, 2026 AT 06:23Jimmy Quilty
March 4, 2026 AT 10:31Miranda Anderson
March 5, 2026 AT 15:06Gigi Valdez
March 5, 2026 AT 15:18