How Drug-Drug Interactions Work: Mechanisms and Effects Explained
- Dorian Wakefield
- 20 Jan 2026
- Pharmacy
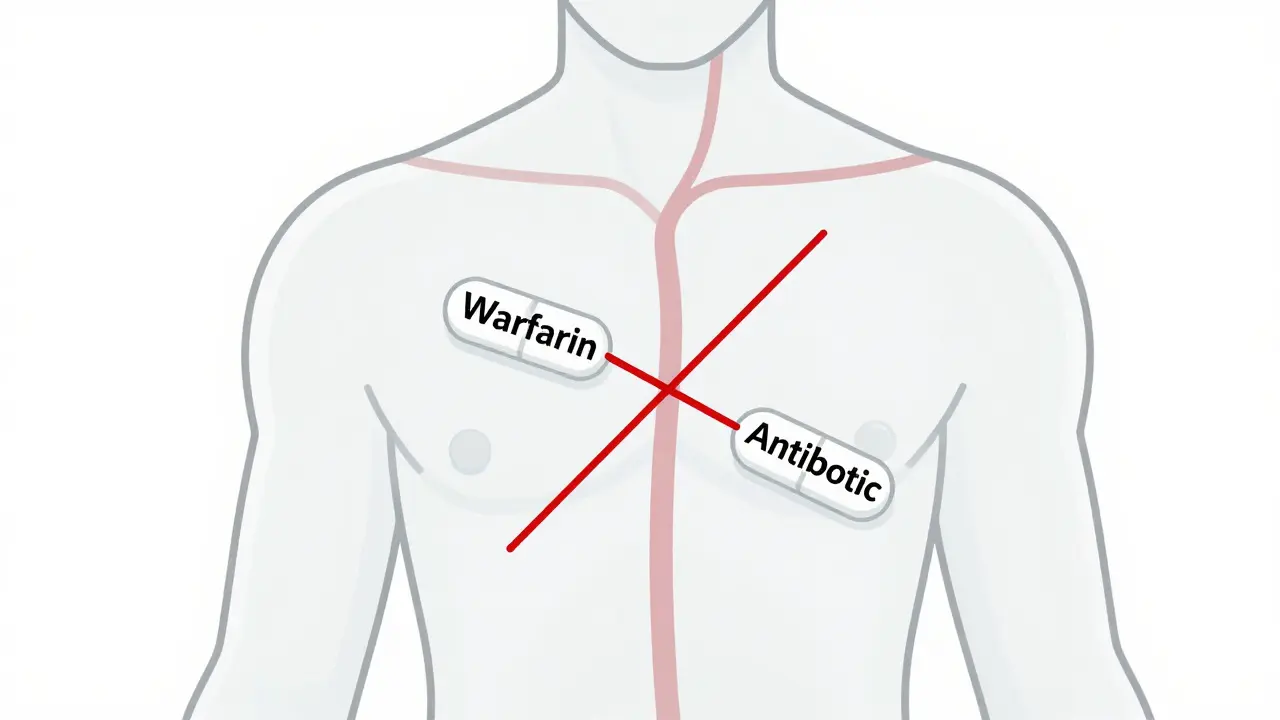
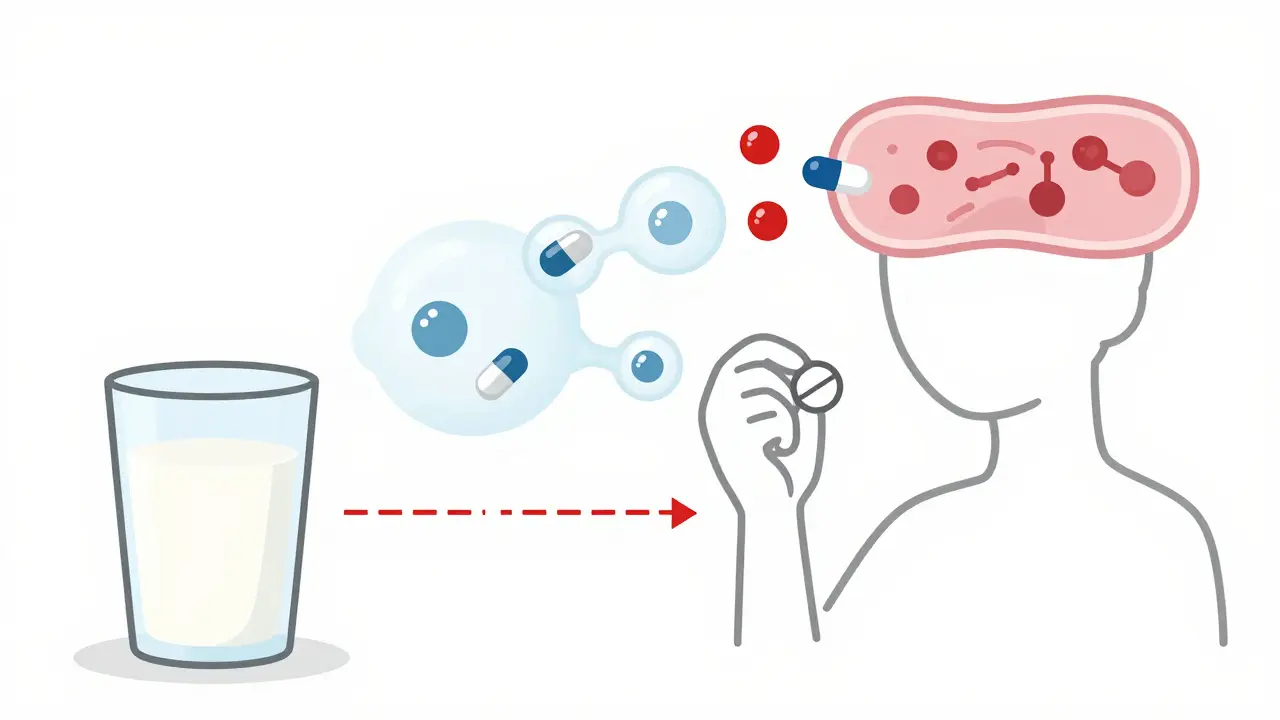
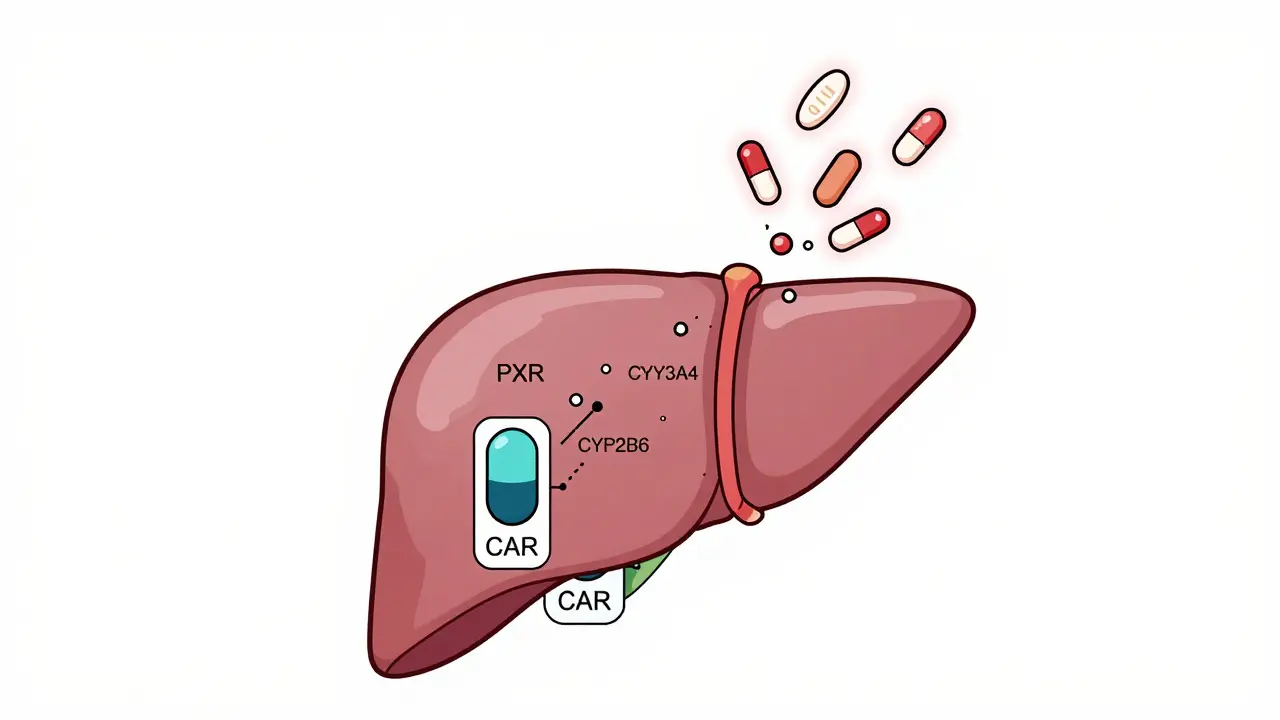
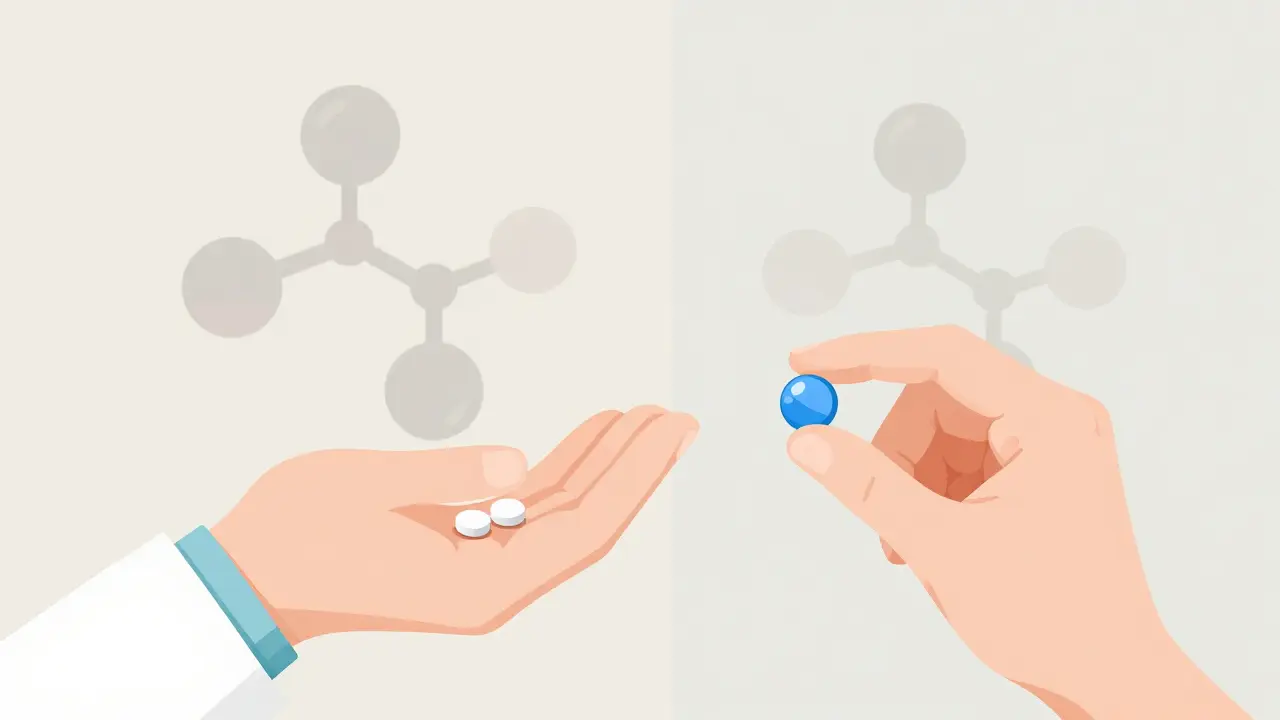
Drug-drug interactions occur when medications affect each other's absorption, metabolism, or effects in the body. Learn how CYP3A4, genetic factors, and common drug combos like warfarin and statins can lead to serious health risks-and how to prevent them.
View More